Medical Devices
-
VC funding in medtech ticks up, raising hopes that slump has bottomed
Analysts also forecast renewed interest in M&A now that large companies have worked through a string of spinoffs.
By Nick Paul Taylor • Aug. 25, 2023 -
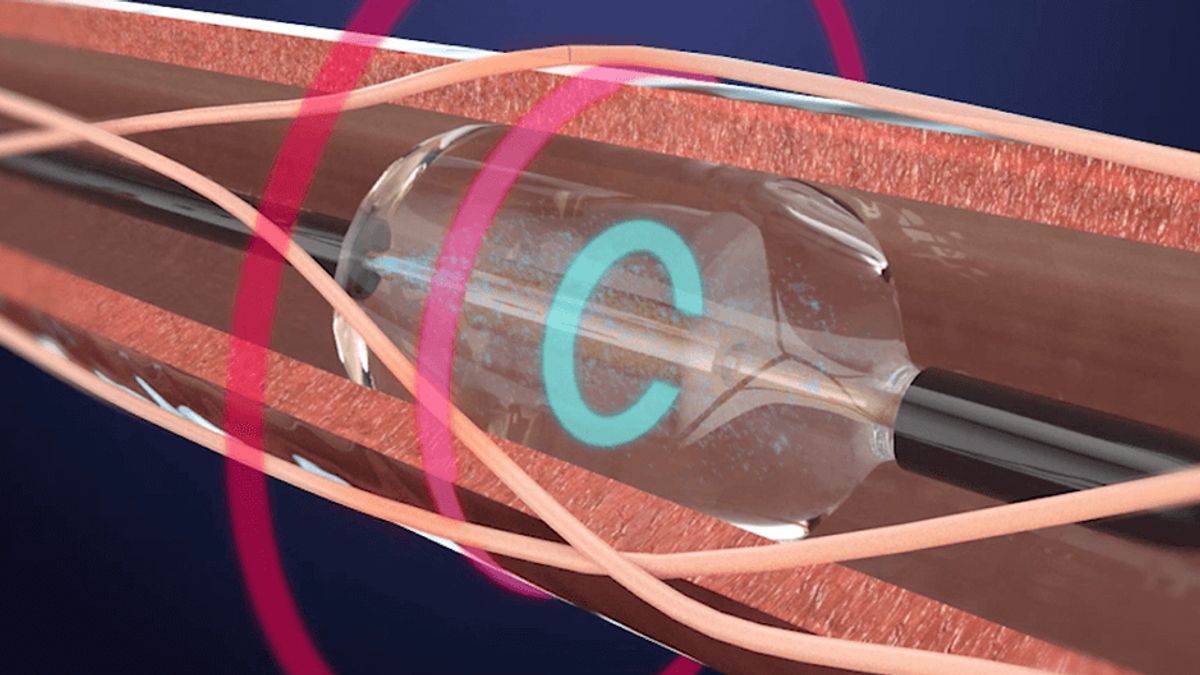
Medtronic’s endovascular artificial intelligence pilot program treats first patient
A physician in the pilot used a 3D map of patient tissue in the image-guided minimally invasive surgery.
By Nick Paul Taylor • Aug. 25, 2023 -
Trendline
The EU Medical Device Regulation
While the European Union’s MDR went into effect in May 2021, device makers still face a range of uncertainties and potential problems with continued extensions and outstanding questions still in play.
By MedTech Dive staff -
‘Business as usual’ at Zimmer Biomet despite sudden CEO change
Incoming Chief Executive Ivan Tornos could accelerate the orthopedics company’s turnaround through acquisitions and by expanding its margins, analysts said.
By Elise Reuter • Aug. 24, 2023 -
Medtronic renal denervation device hits setback at FDA panel
The agency’s outside advisers found the risks of the procedure to lower blood pressure outweighed the benefits in a narrow vote.
By Susan Kelly • Aug. 24, 2023 -
Draeger’s ventilator sound insulation recall triggers FDA Class I notification
The company found concentrations of a potentially carcinogenic foam component were above acceptable levels for children.
By Nick Paul Taylor • Aug. 24, 2023 -
Recor wins FDA panel’s backing for high blood pressure device
The advisers found the company’s renal denervation system is safe and effective, but several panelists expressed concern about the long-term durability of the treatment.
By Susan Kelly • Aug. 23, 2023 -
Bayer inks deal to commercialize Mahana’s prescription digital therapeutics
The multimillion-dollar distribution and marketing partnership comes amid a decline in funding for prescription digital therapeutic companies.
By Nick Paul Taylor • Aug. 23, 2023 -
Levita’s surgeon-controlled arm for magnetic procedures gains 510(k) clearance
The device maker says the technology can reduce the need for an additional assistant in the operating room, driving efficiency.
By Nick Paul Taylor • Aug. 23, 2023 -
Medtronic raises earnings forecast amid supply chain, diabetes business improvements
The company also shared an update on the planned divestment of its monitoring and respiratory interventions segments.
By Elise Reuter • Aug. 22, 2023 -

 Retrieved from Axonics on August 22, 2023
Retrieved from Axonics on August 22, 2023
Axonics appoints Kari Keese to CFO post as Dan Dearen retires
The retirement of company co-founder Dearen is “unexpected but not concerning,” Truist analyst Richard Newitter said.
By Susan Kelly • Aug. 22, 2023 -
Zimmer Biomet CEO Hanson departs for 3M
COO Ivan Tornos will become the orthopedic device maker’s new top executive.
By Elise Reuter • Aug. 22, 2023 -
Bruker to buy PhenomeX and its cell research tools for $108M
The life science tools sector is ripe for consolidation, according to analysts at TD Cowen.
By Susan Kelly • Aug. 18, 2023 -
Deep Dive
Dexcom, Abbott ramp up DTC marketing amid fight for share of growing diabetes market
As insurers cover continuous glucose monitors for more people, device-makers are taking a page from the pharma marketing playbook.
By Elise Reuter • Aug. 17, 2023 -
Integra receives warning letter related to quality issues at Boston plant
The company recalled all products made at its Boston facility over the last five years, and hopes to restart manufacturing in the fourth quarter.
By Elise Reuter • Updated Aug. 17, 2023 -
One death, 2 injuries reported in recall of Philips Trilogy ventilators
Philips warned in a previous recall about problems with the devices’ sensors that can lead to under-delivery of oxygen.
By Elise Reuter • Aug. 16, 2023 -

 Retrieved from GE HealthCare via Business Wire.
Retrieved from GE HealthCare via Business Wire.
GE HealthCare wins FDA clearance for wireless hospital vital sign monitor
The Portrait Mobile device is one of GE HealthCare’s “first major introductions in monitoring in recent years,” according to the company.
By Nick Paul Taylor • Aug. 16, 2023 -
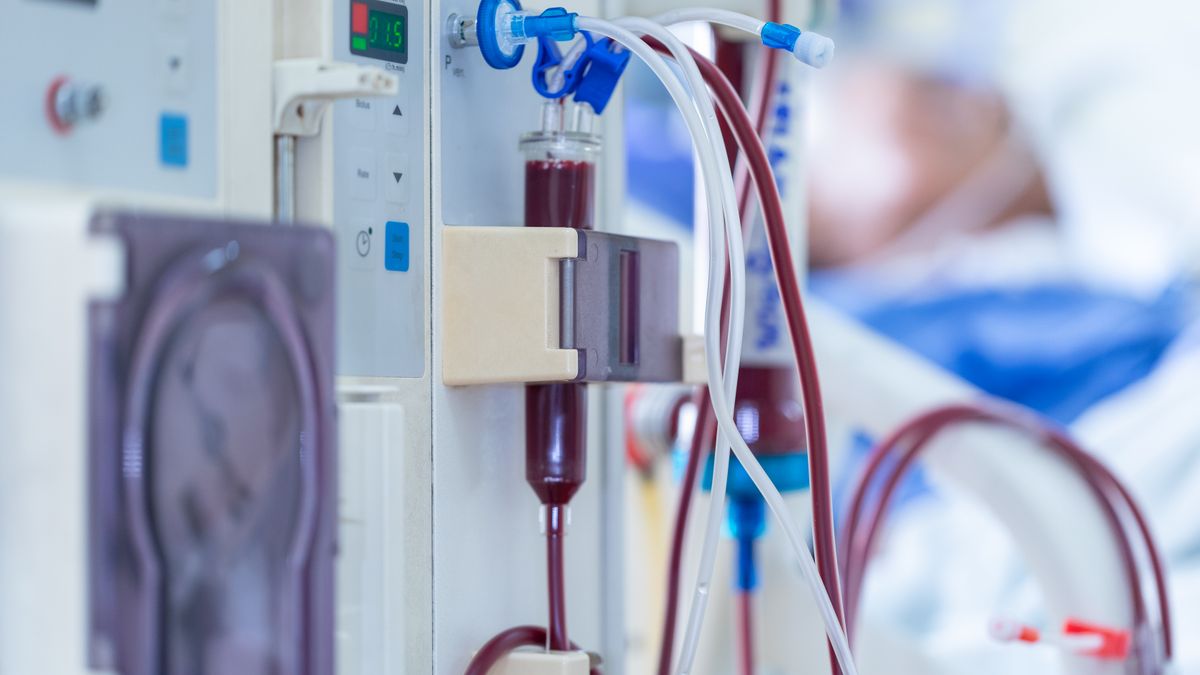
Medtronic’s dialysis device recall assigned to Class I by the FDA
The company contacted customers after routine manufacturing testing identified a blockage that could obstruct the catheter.
By Nick Paul Taylor • Aug. 16, 2023 -
Insulet claims EOFlow copied patch-pump design in patent suit
Insulet filed the lawsuit as competitor Medtronic plans to buy EOFlow for $738 million.
By Elise Reuter • Aug. 15, 2023 -
Medtronic’s bid to dismiss heart device kickback claims fails, clearing case to advance
A whistleblower accused Medtronic of violating the False Claims Act by billing the government for unnecessary medical procedures.
By Nick Paul Taylor • Updated Aug. 15, 2023 -
Boston Scientific’s pivotal pulsed field ablation trial expected to succeed: analysts
Positive results could position Boston Scientific to win U.S. approval for Farapulse in the second half of next year.
By Nick Paul Taylor • Aug. 14, 2023 -
Senseonics seeks iCGM designation for implantable glucose monitor
The company also plans to submit results of a pivotal trial for a version of its device that can be worn for a year.
By Elise Reuter • Aug. 11, 2023 -
Median lag between FDA authorization and Medicare coverage hits 6 years: study
Fewer than 30% of novel medical devices obtain some Medicare coverage in the three years after regulatory authorization, researchers found.
By Nick Paul Taylor • Aug. 11, 2023 -
Getinge’s heart assist pump shutdown problem given FDA Class I recall tag
The company notified healthcare providers after receiving 26 complaints about Cardiosave devices, used to support severely ill patients, unexpectedly shutting down.
By Nick Paul Taylor • Aug. 11, 2023 -
Boston Scientific receives FDA approval for cryoablation system
The company hopes to build on interest in Europe and Japan as competitors bring new cardiac ablation devices to market.
By Elise Reuter • Aug. 10, 2023 -
ZimVie wins FDA approval for smaller Mobi-C cervical disc
The device maker said the new product will “address the anatomical needs of the U.S. patient population.”
By Nick Paul Taylor • Aug. 10, 2023